The Eight Domains of Adaptive Resilience
How to put “the best approximation of truth humans possess” to the test
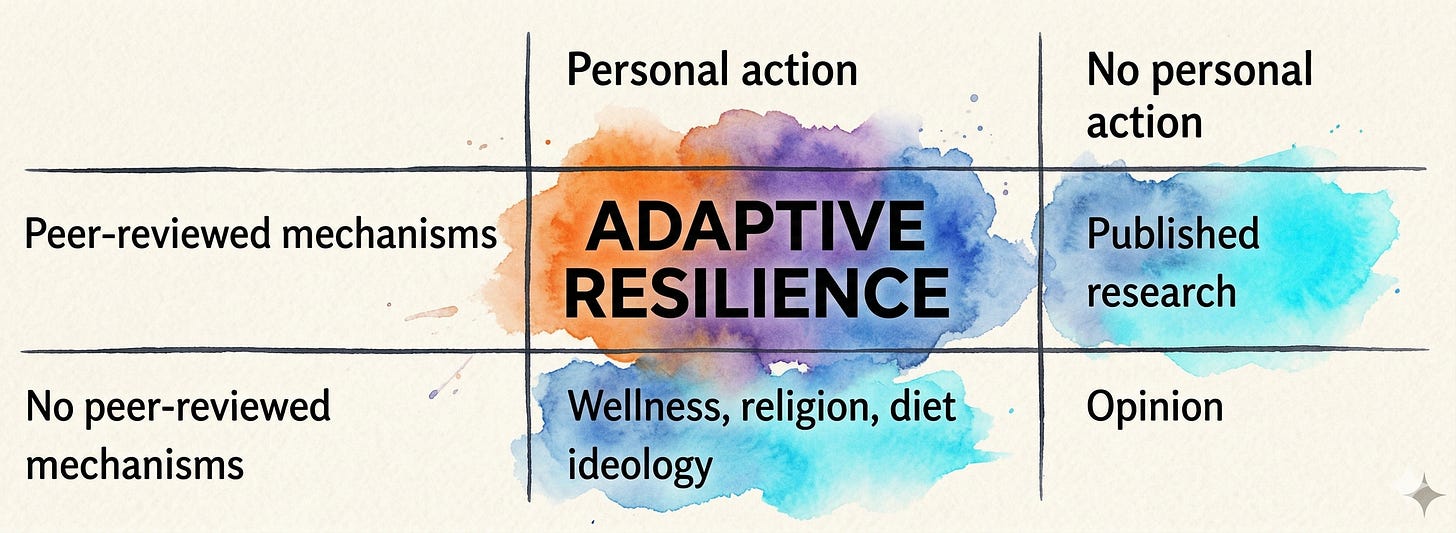
Adaptive Resilience is the systematic, evidence-based realignment of behavior, environment, and community with the biological conditions under which people flourish.
People evolved for specific conditions: small-band, high-trust interdependence. Circadian and seasonal entrainment. Diverse plant-rich diets co-evolved with gut microbiota. Acute stress followed by deep recovery. Embodied movement across varied terrain. Nature contact. Awe through scale and complexity.
Modern environments violate nearly every one of these conditions. The result is not personal failure. It is ‘systems mismatch.’
This practice is grounded in peer-reviewed science — the best approximation of truth we have. It takes that science and puts it into personal action. Where cultural practices, dietary ideologies, or institutional traditions produce measurable benefits, this practice identifies the active behavioral mechanism. Where they do not, they are not included.
Structural conditions like financial precarity, housing instability, and environmental toxin exposure produce measurable inflammatory, cardiovascular, and cognitive consequences. This plan does not prescribe interventions for these conditions because the peer-reviewed evidence base does not support specific behavioral doses the way it does for sleep, movement, or nutrition. But these conditions constrain the biology this plan acts on. Acknowledging that boundary is part of the rigor.
AI is foundational to this practice. People outsource decisions to experts constantly — doctors, nutritionists, advisors — but in an extractive system, those experts operate inside incentive structures that do not always align with your flourishing. AI gives people direct access to the peer-reviewed evidence base, the ability to interpret it, and the capacity to apply it personally. The bottom-right quadrant of this table — opinion without evidence or action — becomes obsolete when anyone can interrogate the literature directly. AI does not replace judgment. It upgrades the inputs to judgment.
The practice keeps updating. That is the point.
1. Rhythm and Recovery
The science of sleep regularity and circadian health is anchored at two institutions on opposite sides of the world. At Monash University in Melbourne, Associate Prof. Andrew Phillips and Prof. Sean Cain — then at the Turner Institute for Brain and Mental Health, now both at Flinders University in Adelaide, South Australia — developed the Sleep Regularity Index (SRI) and produced the landmark studies showing that sleep regularity predicts mortality more strongly than sleep duration — work done in collaboration with Prof. Richa Saxena and Dr. Jacqueline Lane at the Broad Institute and Brigham and Women’s Hospital at Harvard. The UK Biobank, a prospective cohort of over 500,000 adults recruited across 22 assessment centers in the United Kingdom, provides the population data underlying these findings. At the Salk Institute for Biological Studies in La Jolla, California, Prof. Satchin Panda’s lab established the circadian biology of peripheral organ clocks — the discovery that the gut, liver, and other organs maintain their own 24-hour rhythms, synchronized to feeding and light. At the University of Oxford in England, Prof. Russell Foster CBE FRS and his Sleep and Circadian Neuroscience Institute studies how light exposure patterns entrain the master clock in the suprachiasmatic nucleus. The convergence of these groups — Australian chronobiology (the Phillips-Cain group migrated from Monash in Melbourne to Flinders in Adelaide, a brain-drain shift worth tracking), British population data, American circadian molecular biology, and Oxford neuroscience — has reshaped how we understand sleep: not as passive rest, but as the body’s primary infrastructure for repair, immune regulation, and metabolic coordination.
The Practice:
1.1 Wake and sleep at consistent times. Regularity over duration. The SRI data is unambiguous: the single most protective sleep behavior is going to bed and waking up at the same time every day, including weekends. At the optimal tier, the target is an SRI above the 80th percentile — the range where mortality risk plateaus at its lowest.
1.2 Morning light exposure within 30 minutes of waking. Bright, full-spectrum light — ideally direct sunlight — is the strongest zeitgeber (time-giver) for the circadian system. Evening low-light wind-down beginning 2 hours before sleep. The spectral composition matters: blue-enriched light suppresses melatonin; amber and red wavelengths do not.
1.3 Evening reset: gentle movement, warm shower, digital sunset. Screens off or filtered to amber. The body’s core temperature drop is a prerequisite for sleep onset — a warm shower paradoxically accelerates this by driving vasodilation and subsequent heat dissipation.
1.4 No food 3–4 hours before sleep. The digestive system has its own circadian clock. Late eating desynchronizes it from the master clock, impairing overnight metabolic processes. An eating window that closes well before sleep respects the circadian architecture of digestion (see Domain 4.3).
2. Nutrition and Collective Ecology
The science of the gut microbiome and nutrition is concentrated in a handful of institutions doing transformative work. At UC San Diego in La Jolla, California, Prof. Rob Knight’s lab — together with co-founders Dr. Jack Gilbert and Jeff Leach, and scientific director Dr. Daniel McDonald — built the American Gut Project (now the Microsetta Initiative), the world’s largest citizen-science microbiome study, with over 10,000 participants from 40+ countries. Their 2018 publication in mSystems established the 30-plant-per-week finding that anchors this domain’s primary dietary target. At King’s College London in England, Prof. Tim Spector runs the TwinsUK cohort and the British Gut Project, producing parallel evidence on how dietary patterns shape microbial communities in a European population. At the University of Jena in Thuringia, Germany, Lu Zhang (then a PhD candidate, thesis defended March 2026) and colleagues in the Microbiome Dynamics group led by Prof. Dr. Gianni Panagiotou at the Leibniz Institute for Natural Product Research and Infection Biology (Leibniz-HKI) and the Cluster of Excellence “Balance of the Microverse” produced the 2025 Nature Microbiology paper mapping phytonutrient biotransformation across 3,000+ global microbiomes — the most comprehensive study to date of how gut bacteria chemically transform what you eat. And at Tufts University in Medford, Massachusetts, Prof. Michael Levin’s work on biological agency and collective intelligence at every scale of organization provides the theoretical framework this domain draws on: the microbiome is part of you and your collective intelligence, not a passenger.
The Practice:
2.1 High-fiber, plant-diverse meals. 30+ distinct “Outer Holozoa” plant and mushroom species per week as a microbial diversity target — but at the optimal tier, aim higher. This includes herbs, spices, seeds, nuts, grains, legumes, fruits, vegetables, and mushrooms. Every distinct species provides a different fiber profile and phytonutrient substrate. The microbial diversity signal scales with plant and fungi diversity, not total volume.
2.2 Whole foods. Minimize ultra-processed intake. Ultra-processed food is a microbial extinction event — it strips the substrates the ecology depends on and replaces them with compounds that favor low-diversity, pro-inflammatory microbial communities.
2.3 Hydration and mineral balance. Electrolytes matter, especially with high-fiber diets and movement. Magnesium, potassium, and sodium all participate in gut motility and cellular hydration.
2.4 Fermentation as daily practice. Tepache, kombucha, preserved lemons, shrubs, lacto-ferments, kimchi, sauerkraut. Fermented foods deliver both live microorganisms and their metabolic products directly to the gut. A 2021 Stanford RCT (Sonnenburg lab) found that a high-fermented-food diet increased microbial diversity and decreased inflammatory markers more effectively than a high-fiber diet alone over 10 weeks. You are feeding an ecology, not a body.
3. Movement and Capacity
This domain draws on research from two major clusters. In Copenhagen, Denmark, Prof. Bente Klarlund Pedersen MD MDSc at the Centre for Physical Activity Research (CFAS) at Rigshospitalet coined the term “myokines” circa 2003 and established skeletal muscle as an endocrine organ — the foundational insight that exercise is not just biomechanics but biochemical signaling. Her group, and the broader Copenhagen cluster including Prof. Camilla Scheele at the Novo Nordisk Foundation Center for Basic Metabolic Research at the University of Copenhagen, continues to map the muscle-brain axis and muscle-organ crosstalk. In Adelaide, Australia, the Alliance for Research in Exercise, Nutrition and Activity (ARENA) at the University of South Australia in Adelaide has produced the definitive exercise-mortality meta-analyses: Dr. Ben Singh et al.’s 2024 cardiorespiratory fitness meta-analysis (3.8 million observations, 42 studies) and Shailendra et al.’s 2022 resistance training mortality meta-analysis. Dr. Haruki Momma at Tohoku University in Sendai, Japan produced the parallel BJSM meta-analysis on muscle-strengthening activities and chronic disease. The Cleveland Clinic cohort in Cleveland, Ohio (Mandsager et al. 2018, 122,007 patients) anchors the VO2max-mortality dose-response curve. The telomere-exercise link comes from Clodagh Ryall and Dr. Joshua Denham at the University of Southern Queensland in Toowoomba, Queensland, Australia (2025, Journals of Gerontology). Australia and Denmark are producing a disproportionate share of the primary data on why movement extends life — cultures where outdoor activity and cycling remain embedded in daily infrastructure.
The practice:
3.1 VO2max training as a primary longevity target. The goal is elite-tier cardiorespiratory fitness for your age — top 2–5%, or top quartile of a decade younger. This requires dedicated high-intensity interval work (e.g., 4×4 protocol: 4 minutes near-maximal effort, 3–4 minutes recovery, repeated 4 times) at least once per week, in addition to Zone 2 volume.
3.2 Zone 2 aerobic volume. 180–240+ minutes per week. Conversational pace, nasal breathing. Walking, cycling, swimming, rowing. This is the mitochondrial density and fat oxidation base. It also drives steady-state myokine signaling and telomerase activation. More is better; the dose-response curves for cancer, diabetes, and cardiovascular mortality are linear with no observed ceiling.
3.3 Strength training across fundamental patterns: push, pull, squat, hinge, carry. 2–3 sessions per week. The mortality benefit is additive with aerobic work and independent of it. Prioritize progressive overload and compound movements.
3.4 Power training. Include explosive, velocity-dependent movements: jumps, throws, Olympic lift derivatives, fast-tempo loaded movements. Power declines ~3.5% per year after 65 — roughly twice the rate of strength. Training it is not optional for long-term functional independence.
3.5 Incidental movement woven into daily structure. 10,000+ steps as a floor, not a target. Walking as baseline cognition support. Standing, stretching, stair-climbing as defaults. The dose-response for steps and cancer, diabetes, and depressive symptoms is linear — there is no point where more stops helping.
3.6 Flexibility, balance, and mobility work. The body expects varied movement across planes and ranges, not repetitive isolated patterns. This preserves the movement vocabulary that prevents falls, compensatory injury, and progressive loss of functional capacity.
4. Hormesis and Adaptation
The science of hormetic stress spans three geographic clusters. The heat exposure evidence is dominated by Prof. Jari Laukkanen’s group at the University of Eastern Finland in Kuopio, Finland, who run the Kuopio Ischemic Heart Disease cohort — 2,315 men followed for over 20 years — the only long-duration sauna mortality dataset in existence. Dr. Setor Kunutsor at the University of Leicester in England does the meta-analytic and interaction analyses of that same Finnish data. Dr. Vienna Brunt, now at Appalachian State University in Boone, North Carolina, produced the key mechanistic work on passive heat therapy and cardiovascular function at the University of Oregon in Eugene. For cold exposure, Dr. Susanna Søberg completed her PhD at the Center of Inflammation and Metabolism at Rigshospitalet, University of Copenhagen, Denmark, and her 2021 Cell Reports Medicine paper on winter swimmers — with senior author Prof. Camilla Scheele at the Novo Nordisk Foundation Center for Basic Metabolic Research — is the most cited human study on cold adaptation and brown adipose tissue. Dr. Denis Blondin at the Université de Sherbrooke in Sherbrooke, Quebec, Canada leads the mechanistic work on cold-induced thermogenesis and substrate metabolism. For breathing and autonomic regulation, Prof. David Spiegel’s lab at Stanford University School of Medicine in Stanford, California produced the 2023 cyclic sighing RCT. For time-restricted eating, Prof. Satchin Panda at the Salk Institute in La Jolla established the circadian biology of peripheral organ clocks. Finland owns the heat data. Copenhagen owns the cold data. The US contributes the autonomic and circadian mechanisms.
The practice:
4.1 Heat exposure calibrated to core temperature. The biological target is a core body temperature of ≥39°C (102.2°F). This is the threshold for robust heat shock protein activation — HSP70 (protein refolding and cellular protection), HSP90 (initiator of the HSP expression cascade), and HSP27 (cytoprotection). Below this threshold, cardiovascular and endothelial benefits occur but molecular proteostasis signaling is incomplete. In vitro studies on human white blood cells showed that 38.5°C was insufficient for HSP induction in monocytes, while 39°C produced significant HSP70 upregulation. Hot water immersion protocols targeting 39.5°C for 60 minutes produced significant increases in both HSP27 and HSP70. Heated exercise protocols reaching 39.5°C increased HSP90, the initiator of the entire cascade.
Dry sauna at 80–100°C for 30–40 minutes is a typical protocol to reach the 39°C core threshold, but individual variation is significant — body composition, hydration, acclimation history, and ambient humidity all matter. Core temperature measurement (ingestible thermometry pill, or calibrated tympanic probe) is the only way to verify you’ve arrived. Sweat is not the signal. Internal temperature is.
Heat acclimation attenuates the HSP response. Studies show that by the tenth sauna session at a fixed temperature, core temperature was limited to 38.2°C and the HSP response was largely eroded. Thermal stress, like mechanical stress, requires progressive overload — longer durations, higher temperatures, or reduced cooling between sessions.
Frequency: 4–7 sessions per week. The Finnish Kuopio Ischemic Heart Disease cohort (2,315 men, 20.7 years follow-up) found that men who used sauna 4–7 times per week had 63% lower risk of sudden cardiac death, 48% lower risk of fatal CVD, 50% lower all-cause mortality, and 66% lower risk of dementia compared to once-weekly users. A 2024 analysis of the same cohort found that frequent sauna use counteracted the adverse mortality effects of elevated blood pressure — suggesting heat exposure is particularly protective under existing cardiometabolic stress.
4.2 Cold exposure for autonomic training and neurochemical regulation. Cold water immersion (≤15°C) triggers an acute catecholamine surge — norepinephrine increases 200–300% and dopamine approximately 250% from a single exposure, with effects lasting two to three hours. These magnitudes exceed most pharmacological interventions for alertness and mood. Cold also activates brown adipose tissue thermogenesis, improves insulin sensitivity, and induces cold shock proteins (particularly RBM3) that enhance cellular resilience. Søberg et al. (2021, Cell Reports Medicine, University of Copenhagen) found that experienced winter swimmers who combined cold water immersion with sauna showed dramatically enhanced cold-induced thermogenesis — 500–1,000 kcal/24h during cooling versus approximately 20 kcal/24h in controls — despite similar brown fat glucose uptake, suggesting improved thermoregulatory efficiency rather than simply more brown fat.
A 2025 systematic review and meta-analysis of randomized trials found that cold water immersion modulates neurobiological markers associated with mental health: reduced cortisol, increased norepinephrine, and reduced pro-inflammatory cytokines including IL-6 and TNF-α. A separate 2025 systematic review confirmed the acute physiological effects are real and measurable — not placebo.
However, the evidence base for cold is younger and less robust than for heat. The long-term mortality data that exists for sauna (Finnish cohort, 20+ years) does not yet exist for cold. The mental health and metabolic evidence is promising but drawn from small, short-duration trials. What is strong: the acute neurochemical and autonomic effects. What is emerging: the metabolic and cellular resilience effects. What is absent: long-term mortality data.
Critical timing constraint: A 2024 meta-analysis in the European Journal of Sport Science confirmed that cold water immersion immediately following resistance training attenuates muscle hypertrophy. CWI blunts mTORC1 signaling, satellite cell activation, and type II fiber growth. This is a direct conflict with Domain 3. If you are training for strength, power, and muscle mass — and this plan says you should be — cold exposure must be temporally separated from resistance training by at least 4–6 hours, or performed on non-training days. Cold after aerobic work does not show the same interference.
Protocol: 2–5 minutes at 10–15°C (50–59°F), full immersion to neck. Start with 30–60 seconds and build. The autonomic training effect — the capacity to remain calm under acute sympathetic activation and recover rapidly into parasympathetic dominance — is itself the adaptation. The goal is not to endure cold. It is to train the shift.
4.3 Time-restricted eating. Align the eating window with circadian biology. The gut has its own peripheral clock. Late eating disrupts it, impairing glucose tolerance, lipid metabolism, and overnight repair processes. An eating window of 8–10 hours, ending 3–4 hours before sleep, respects the circadian architecture of digestion.
4.4 Breathing practices as autonomic inputs. Tummo, box breathing (4-4-4-4), alternate nostril breathing, physiological sigh. These are not rituals. They are inputs to a trainable autonomic system — each pattern drives a specific ratio of sympathetic to parasympathetic activation. The physiological sigh (double inhale through nose, long exhale through mouth) is the fastest known real-time downregulator of sympathetic arousal, validated in a 2023 Stanford RCT (Balban et al., Cell Reports Medicine, Spiegel lab).
5. Nervous System Regulation
The science in this domain is advancing rapidly. The key research groups: Prof. Maiken Nedergaard MD DMSc (University of Rochester Medical Center in Rochester, New York / University of Copenhagen in Denmark) originally defined the glymphatic system in rodents. Dr. Juan Piantino (Oregon Health & Science University in Portland, Oregon) produced the first proof of glymphatic flow in living human brain tissue (2024). Keating (Vanderbilt University in Nashville, Tennessee) and Dr. David Vago (Brigham and Women’s Hospital / Mass General Brigham in Boston, Massachusetts) demonstrated that meditation modulates cerebrospinal fluid dynamics in humans (2025), with Prof. Manus Donahue at Vanderbilt as corresponding author. Hauglund et al. (University of Copenhagen, Denmark) identified the norepinephrine-vasomotion mechanism driving glymphatic clearance during sleep (2025). Prof. David Spiegel (Stanford) ran the RCT establishing cyclic physiological sighing as an autonomic intervention (2023). The field of autonomic regulation and brain waste clearance is converging from multiple directions simultaneously.
The practice:
5.1 Daily breathwork for autonomic control. Box breathing (4-4-4-4), alternate nostril, humming (bhramari), physiological sigh (double inhale through nose, long exhale through mouth). 5–10 minutes. The physiological sigh is the fastest known real-time downregulator of sympathetic arousal — a 2023 Stanford RCT (Balban et al., Cell Reports Medicine, Spiegel lab) found 5 minutes of daily cyclic sighing improved mood and reduced respiratory rate more effectively than mindfulness meditation or box breathing.
5.2 Meditation or focused attention practice. 20–30 minutes. Silent, self-guided breath awareness. The mechanism is CSF flow modulation and glymphatic support — reduced backward flow through the cerebral aqueduct, increased low-frequency oscillations consistent with waste clearance. This effect does not appear from casual attention, guided audio, or breathing rate reduction alone (Vanderbilt PNAS 2025). Sitting, walking, or body scan formats all qualify, but the attentional component is what drives the neurofluid response.
5.3 HRV as a trainable biomarker. Track morning HRV (RMSSD) as a daily readout of autonomic flexibility and recovery status. HRV-guided training — adjusting exercise intensity based on autonomic readiness — is supported as at least as effective as predefined programs. The goal is not a single high number but an upward trend in the 7-day rolling coefficient of variation. Autonomic flexibility — the range of the system — is what predicts resilience.
5.4 Interoceptive check-ins. Pause and assess nervous system state 2–3 times daily. Name it: activated, settled, collapsed. Naming is the first step of regulation. The capacity to detect and label autonomic state is itself a trainable skill that improves regulatory speed.
6. Mental and Cognitive Growth
The science of cognitive reserve, neuroplasticity, and BDNF-mediated learning draws from a dispersed but interconnected research network. At Harvard Medical School and Brigham and Women’s Hospital in Boston, Massachusetts, Dr. Kirk Daffner’s Laboratory of Healthy Cognitive Aging ran the SAGE trial (Successful Aging and Enrichment) — the randomized study establishing that cognitive training independently augments BDNF and mediates cognitive gains, conducted jointly with Linnaeus University in Växjö, Sweden. Dr. Krister Håkansson, now at Karolinska Institutet in Stockholm, Sweden, produced the exercise-before-learning BDNF priming study (2020, Scientific Reports). The cognitive reserve framework originates from Prof. Yaakov Stern at Columbia University in New York City. The 2024 Lancet Commission on dementia — the most authoritative global synthesis of modifiable dementia risk factors — is led by Prof. Gill Livingston at University College London in England, synthesizing evidence from dozens of research groups worldwide. The hippocampal volume and language learning evidence comes from Prof. Christos Pliatsikas (promoted to Professor of Cognitive Neuroscience, August 2025) at the University of Reading in Berkshire, England and collaborators across European universities. The MTT24.5 novelty-learning pilot RCT comes from Buenos Aires. The field is genuinely global — but the BDNF mechanism, the cognitive reserve theory, and the dementia prevention framework are anchored in Boston, Stockholm, London, and New York.
The practice:
6.1 Deep work sessions. Focused learning or creation in blocks of 60–90 minutes. Protect these from interruption. The prefrontal cortex requires sustained uninterrupted engagement to enter the consolidation patterns that produce long-term learning. Fragmented attention does not substitute for depth.
6.2 Sequence movement before learning. When possible, schedule cognitively demanding work after physical exercise — even a 20-minute walk or Zone 2 session. The acute post-exercise BDNF window primes the brain for plasticity. The measured effect in controlled trials is small per session, but this is a daily structural decision that compounds. Over years, the question is not whether one session matters, but whether thousands of optimally sequenced sessions build more reserve than thousands of unsequenced ones. The mechanism says yes.
6.3 Prioritize genuine novelty. Learn something you do not already know how to do. A new language. A musical instrument. An unfamiliar domain of science or craft. The neuroplastic signal from novelty and complexity exceeds the signal from practicing existing skills. Repetition maintains; novelty builds. At the optimal tier, this means sustained engagement with at least one domain that is genuinely outside your current competence at all times.
6.4 Reflection or journaling. Error-correction at the cognitive level. What happened, what I expected, what the delta means. This is not self-help. It is prediction-error processing — the mechanism by which the brain updates its models. Writing externalizes the process and makes the update explicit.
6.5 Creative production. Make something. Write, build, design, compose, code, grow. The act of producing — not just consuming — engages motor planning, executive function, aesthetic judgment, and error correction simultaneously. Production is higher-bandwidth cognitive load than consumption.
7. Connection and Community
The science of social connection and health is built across continents. At Brigham Young University in Provo, Utah, Prof. Julianne Holt-Lunstad has spent two decades producing the meta-analyses that established loneliness and social isolation as mortality risk factors on par with smoking — work that directly informed the U.S. Surgeon General’s 2023 advisory. At UCLA in Los Angeles, California, Prof. Steve Cole’s Social Genomics Core Laboratory identified the molecular mechanism: loneliness changes which genes immune cells express, shifting the body toward chronic inflammation through what he calls the conserved transcriptional response to adversity (CTRA). The largest single meta-analysis — 90 cohort studies, 2.2 million people — was led by Prof. Yashuang Zhao’s group at Harbin Medical University in Harbin, Heilongjiang Province, northeastern China, published in Nature Human Behaviour in 2023. The dementia-connection link is anchored at University College London in England, where Dr. Andrew Sommerlad and Dr. Naaheed Mukadam study how social participation protects against neurodegeneration across the lifespan. Unlike Domains 3–5, where the primary data clusters in Scandinavia, the social connection literature draws from every continent. The biology of social need is universal, even if the cultural forms vary.
The practice:
7.1 Regular face-to-face contact with close relationships. Frequency matters more than duration. A short daily interaction with a trusted person does more for inflammatory regulation than a long monthly gathering. Prioritize the people you would call in a crisis — those relationships are the load-bearing structure.
7.2 Shared meals or social rituals. Eating together is social bonding infrastructure with deep evolutionary roots. It synchronizes circadian cues, regulates nervous system state through co-regulation, and creates predictable rhythms of contact. Make it regular. Make it frequent.
7.3 Play, humor, or laughter daily. These are not trivial. Laughter downregulates cortisol, increases endorphins, and signals safety to the nervous system — both yours and others’. Play is a social technology for building trust without stakes.
7.4 Reciprocal support. Give help. Ask for help. Both activate oxytocin-mediated bonding pathways. The ask is as important as the give — it signals trust, creates interdependence, and breaks the self-sufficiency pattern that isolation feeds on.
7.5 Invest in community structure. Join, build, or maintain a group that meets regularly around a shared purpose. The specific purpose matters less than the rhythm, the trust, and the mutual accountability. This is the closest behavioral analog to the ancestral band. It does not happen passively. It requires the same intentional investment as any other training practice in this plan.
8. Nature and Environment
The science of nature and health has two geographic epicenters. In Tokyo, Japan, Dr. Qing Li at Nippon Medical School’s Department of Hygiene and Public Health has spent two decades building the field he named Forest Medicine — studying the immune, endocrine, and nervous system effects of shinrin-yoku (forest bathing) in Japan’s designated forest therapy bases. His work on natural killer cell activation and phytoncides is the foundation of the immunological case for nature contact. In the UK, the large meta-analyses quantifying greenspace exposure and health outcomes come from Dr. Caoimhe Twohig-Bennett and Prof. Andy Jones at the University of East Anglia in Norwich, England, and from ISGlobal (Barcelona Institute for Global Health) in Barcelona, Catalonia, Spain, where Dr. David Rojas-Rueda and Prof. Mark Nieuwenhuijsen produced the green space mortality meta-analyses. Japan is to forest bathing what Finland is to sauna — the cultural practice predated the science by generations, and the research emerged from people studying traditions their own population never abandoned.
The practice:
8.1 Daily outdoor time. Walking, gardening, sunlight. Minimum 30 minutes. The cortisol dose-response curve flattens after 20–30 minutes, but the circadian, immune, and microbial benefits accumulate with longer exposure. More is better. At the optimal tier, outdoor time is not a scheduled intervention — it is the default environment for movement (Domain 3), social contact (Domain 7), and cognitive work (Domain 6) whenever conditions allow.
8.2 Forest or green space immersion weekly. The shinrin-yoku research base shows NK cell activation lasting 30+ days from a multi-day forest trip. A weekly two-hour immersion in a forested or biodiverse green space is the minimum to maintain elevated immune function. Breathe through the nose. The phytoncides are airborne. The immune signal enters through the lungs.
8.3 Microbiome exposures. Soil contact, plant handling, composting, animal contact. Hands in dirt is applied microbiology. Gardening specifically combines soil microbiome exposure, sunlight, gentle movement, and sensory engagement — it touches Domains 2, 3, 4, and 8 simultaneously. It is one of the highest-density behaviors in this plan.
8.4 Minimize environmental toxins. Air quality, water quality, light pollution. The environment shapes the biology — it always has. Indoor air in sealed buildings can be 2–5 times more polluted than outdoor air. Blue-enriched artificial light at night disrupts melatonin and circadian architecture (Domain 1). Municipal water quality varies. These are not paranoid concerns. They are environmental inputs that constrain the biology this plan acts on, and they are modifiable.
The Daily Practice, In short
Morning:
Wake up early, within 30 minutes of yesterday
Morning sunlight within 30 minutes of waking
Breath work or meditation
Zone 2 cardio
Strength training
Flexibility, balance, and mobility
Day:
Sauna and cold exposure
Eat within a window of time, during the day
Eat dozens of diverse whole foods outside of Holozoa — plants, fungi, ferments
Walk and move throughout the day
Focused deep work sessions and skill-building
Laugh and play
Connect with someone you trust
Spend time outdoors, hands on soil or plants
Evening:
Dim lights at night, screens off
Journal or reflect
Evening wind-down: move, shower, no screens
Sleep within 30 minutes of yesterday
Night:
Deep rest
Recently, I marked the anniversary of an accident that changed my life. As a child, I experienced trauma broke my spine and shattered my heel bone into more than 42 pieces.
Doctors told my mother that I might not be able to walk again normally. (Luckily for me, she never told me that part.)
A reminder that ‘perseverance’ is better with a little help. How inspiring is it when people tell you what you can do instead of what you can’t?
As I continue to work on my philosophy and practice, both the tools and the science are continually improving. Today, I look back, and have clocked a few thousand kilometers running by the beach in Alameda; and I have no reason to believe my traumatic injuries are permanent.



